Cell Transplantation: A Novel Therapeutic Modality for Cerebral Palsy with Co-Morbid Intellectual Disability
Alok Sharma, Hemangi Sane, Nandini Gokulchandran, Pooja Kulkarni, Alitta Jose, Vishal Ganar, Samson Nivins and Prerna Badhe
DOI10.21767/2471-8548.10010
Alok Sharma1, Hemangi Sane2, Nandini Gokulchandran1, Pooja Kulkarni2, Alitta Jose2*, Vishal Ganar3, Samson Nivins2 and Prerna Badhe4
1Department of Medical Services and Clinical Research, NeuroGen Brain and Spine Institute, India
2Department of Research and Development, NeuroGen Brain and Spine Institute, India
3Department of Neurorehabilitation, NeuroGen Brain and Spine Institute, India
4Department of Regenerative Laboratory Services, NeuroGen Brain and Spine Institute, India
- *Corresponding Author:
- Alitta Jose
Department of Research and Development
NeuroGen Brain and Spine Institute, India
Tel: +9122-41136565
E-mail: publications@neurogen.in
Received Date: August 27, 2018; Accepted Date: December 14, 2018; Published Date: December 21, 2018
Citation: Sharma A, Sane H, Gokulchandran N, Kulkarni P, Jose A, et al. (2018) Cell Transplantation: A Novel Therapeutic Modality for Cerebral Palsy with Co-Morbid Intellectual Disability. J Neuropsychiatry Vol.2 No.2:6. DOI: 10.21767/2471-8548.10010
Copyright: © 2018 Sharma A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Cerebral palsy (CP) is a non-progressive disorder. The global incidence of neonatal CP is 1-5 per 1000 live births where 45% of children with CP have a comorbid intellectual disability (ID). This affects the quality of life and reduces life expectancy of affected individuals.
Methods and findings: With an aim to study the efficacy and safety of autologous bone marrow mononuclear cells (BMMNCs) transplantation in CP with ID, we administered a 17-year-old boy with BMMNCs along with neurorehabilitation. On follow up after 6 months, clinical improvements were recorded in speech, fine motor skills, ambulation, oromotor skills, social awareness, cognitive skills, memory and attention. Comparative Positron Emission Tomography- Computer Tomography scan of brain before and six months after cell transplantation revealed improved metabolism in the anterior cingulate cortex, left caudate head, thalamus, medial temporal cortex, cerebellum. Intelligent Quotient improved from 21 to 29. Social age according to the Vineland Social Maturity Scale was increased by 1 year. Functional Independence Measure and Gross Motor Functional Measure increased from 22 to 26 and 74.65 to 76 respectively. On Gross Motor Functional Classification System, he improved from IV to III.
Conclusion: These changes suggest that BMMNCs transplantation along with neurorehabilitation was effective for this child suffering from CP with ID.
Keywords
Cerebral palsy; Intellectual disability; Autologous bone marrow mononuclear cells; Cell transplantation; Positron emission tomography-computer tomography
Introduction
Cerebral palsy represents a group of clinically heterogeneous non-progressive disorders of movement and posture causing activity limitation attributed to a static disturbance in the developing brain [1]. The causes are congenital, genetic, inflammatory, infectious, anoxic, traumatic and metabolic. Due to advances in medical field, the cases of CP are rising with increase in survival of very preterm and low birth weight infants. The global incidence of neonatal CP is at 1-5 per 1000 live births [2]. CP, although a motor disorder, has associated neurocognitive and sensory impairments. Among children, the estimated prevalence of cerebral palsy with co morbid ID varies from 30 to 77 percent [3]. This adversely affects the response to treatment and can further compromise the quality of life, reduce life expectancy of affected individuals and increase the burden of care on their families [4].
Despite the various traditional therapeutic modalities, the neurological impairment results into lifelong disabilities. Aim of the available treatment strategies like surgical intervention, medicine, physiotherapy, occupational therapy is only to manage the symptoms [5,6]. There is no definitive cure for CP with ID because of the inability of the neurons to instinctively repair themselves [7]. Hence there is a need for an intervention that addresses the core neuropathology of the brain. Autologous BMMNCs transplantation has been a promising therapeutic modality in different neurodevelopmental disorders due to its neuroprotective and neurorestorative properties [8-10].
To study the effect of cellular transplantation in CP with ID, we administered a 17-year-old patient with autologous BMMNCs, intrathecally.
Functional imaging techniques such as PET CT scan help in detecting metabolic abnormalities and changes in regional blood flow hence, we used the PET CT scan of brain as a monitoring tool to study the effect of cell transplantation at the cellular level [11].
Case Presentation
A 17-year-old boy of 1st degree consanguineous marriage was a full-term normal delivery but did not cry immediately after birth. He had suffered hypoxia and was administered oxygen for 5 minutes. History of neonatal jaundice was present. Fifteen days after birth, he had chronic diarrhea which continued till 2 to 4 years. All motor, mental and speech milestones were delayed. He achieved head control at 4 months whereas rolling, crawling and creeping were missed. Sitting independently and walking with support was achieved at the age of 4 years and speech was monosyllabic at the age of 5 years. The patient was treated for bilateral cataract at the age of 15 years.
Neurologically, he was hypertonic and hyperreflexic. The voluntary control in the lower and upper extremities was fair. Fine motor activities were affected as he was unable to hold anything in his hands. He walked with a diplegic gait with scissoring pattern, crouched posture, foot everted and dorsiflexed. Postural alignment while sitting was kyphotic with poor balance. Oromotor issues like swallowing difficulty and drooling were also present. Cognition, problem solving abilities and higher metal functions were affected. He had aggressive behavior along with temper tantrums and wouldn’t respond to any of the commands. Awareness, orientation, imitation and judgement skills were poor. Eye contact was fleeting with poor vision. Functionally, he required assistance in most activities of daily living (ADLs). His sleep pattern was disturbed, and sensations were also affected. Functional Independence Measure (FIM) score was 22. Gross Motor Functional Measure (GMFM) was 74.65 and Gross Motor Functional Classification System (GMFCS) was at level IV. Intelligence quotient (IQ) test was done to indicate the level of ID which was found to be 21. Social age according to the Vineland Social Maturity Scale (VSMS) was found to be 3 years and 2 months.
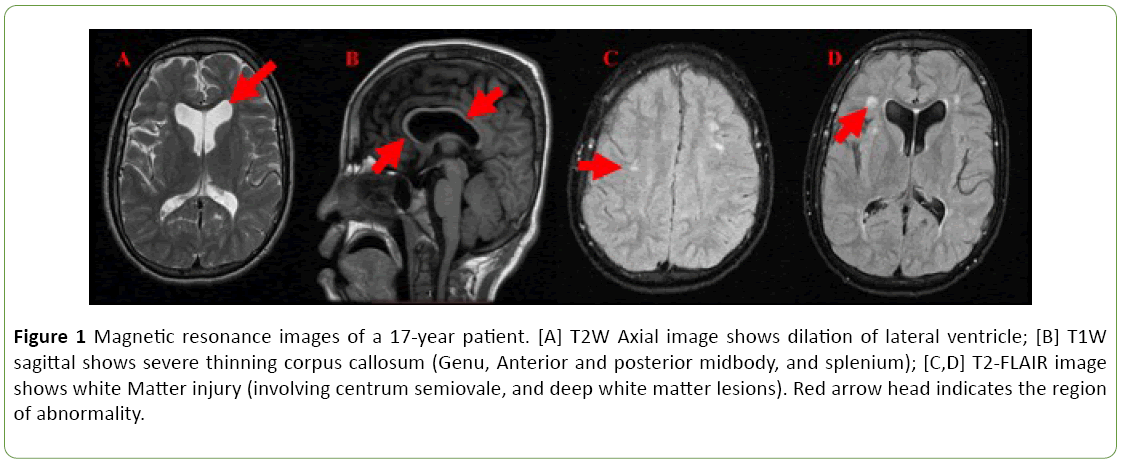
Positron Emission Tomography- Computer Tomography (PET CT) of brain revealed reduced metabolism in anterior cingulate cortex, right temporal cortex, left caudate head, thalamus, medial temporal cortex and cerebellum. Mildly reduced metabolism was observed in superior frontal cortex. Ventricular dilation was prominently seen. BERA and EEG report was found to be normal. MRI brain revealed T2 FLAIR hyperintensities in the centrum semiovale, deep white matter and periventricular regions. Ex-vacuo dilation of lateral ventricles was seen. Severe thinning of corpus callosum including genu, anterior and posterior midbody, and splenium were noted (Figure 1).
Figure 1: Magnetic resonance images of a 17-year patient. [A] T2W Axial image shows dilation of lateral ventricle; [B] T1W sagittal shows severe thinning corpus callosum (Genu, Anterior and posterior midbody, and splenium); [C,D] T2-FLAIR image shows white Matter injury (involving centrum semiovale, and deep white matter lesions). Red arrow head indicates the region of abnormality.
Methodology
Selection of the patient was done based on World Medical Association Helsinki declaration [12]. The protocol was reviewed and approved by Institutional Committee for Stem Cell Research and therapy (IC-SCRT). Parents signed the informed consent form after a detailed explanation of the procedure. A week before the cell therapy, the patient underwent serological, biochemical and hematological blood tests, PET CT scan of brain, EEG, BERA, MRI Brain and chest Xray.
300 mcg of granulocyte colony stimulating factor (GCSF) was administered 72 hours and 24 hours before the cell therapy. The procedure was done under all aseptic conditions in the operation theatre. The patient was laid down in the supine position and the bone marrow was collected from the right anterior superior iliac spine under local anesthesia with sedation. 110 ml of the bone marrow was aspirated using the bone marrow aspiration needle and collected in heparinized tubes. It was then transferred to the laboratory where the Mononuclear cells (MNCs) were separated using a density gradient method. CD34+ count was performed using Fluorescence activated cell sorter (FACS) and the viability was found 98%. 104-106 cells were injected intrathecally into the space between 4th-5th lumbar vertebra in the operating room. Simultaneously, 675 mg methyl prednisolone in 500 ml Isolyte P was given intravenously to manage the immediate inflammation.
Cellular therapy was followed by multidisciplinary neurorehabilitation which included physiotherapy, occupational therapy, speech therapy along with psychological intervention and special education.
After the first cellular therapy, the patient was followed up after 3 months and 6 months to assess the effect of intervention. FIM, GMFCS, GMFM, IQ, VSMS and PET CT were the outcome measures used.
Results
At three months follow up, improvements were noted in the higher mental functions. Improvements were seen in cognitive skills, memory, orientation, attention and concentration of the patient as reflected in his schooling status. Temper tantrums, aggressive behavior, hyperactivity and laughing without reason had reduced. Socially, he started interacting with people and responded to the commands, but eye contact remained poor. Speech improved as he could say one to two words which he couldn’t earlier. Oromotor skills improved as he had no difficulty in swallowing the food and drooling had stopped. Fine motor skills improved as he could hold things now. Sitting, standing balance and posture had also improved. He could walk independently holding the walking frame and high boots for half an hour. FIM score increased from 22 to 25. GMFM score increased from 74.65 to 76.04. On the GMFCS scale, the patient was promoted from level IV to level III. IQ remained the same.
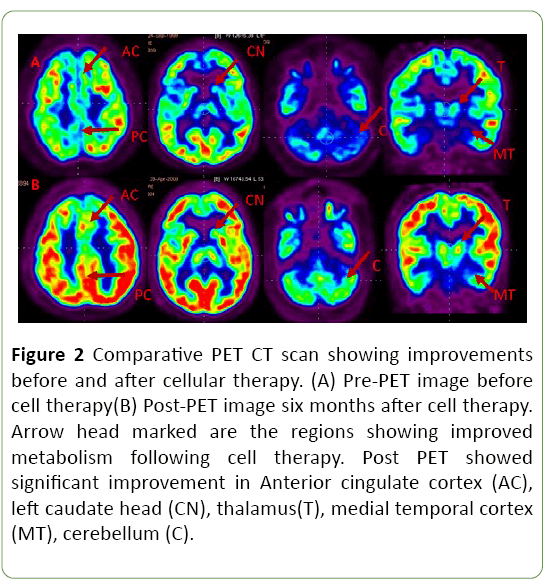
At six months follow-up, his sit to stand transitions improved. Transfers at different levels were much easier and smoother. Voluntary control improved. Fine motor skills were better than before as he could hold objects for longer time in his hand. He could hold a pen for some time and draw sketches. His walking balance improved, and he did not put his body weight on others. Dependency to perform ADLs was minimal. Further improvements were seen in his higher mental functions. At school, his teachers reported improvement in the educational status and peer interaction. IQ range improved from 21 to 29. Social age according to the Vineland Social Maturity Scale (VSMS) was found to be 4 years and 4 months whereas 6 months earlier it was 3 years 2 months. FIM score improved from 25 to 26. A repeat PET CT scan of the brain when compared to preintervention scan showed improved metabolism in anterior cingulate cortex, left caudate head, thalamus, medial temporal cortex, cerebellum (Figure 2).
Figure 2: Comparative PET CT scan showing improvements before and after cellular therapy. (A) Pre-PET image before cell therapy(B) Post-PET image six months after cell therapy. Arrow head marked are the regions showing improved metabolism following cell therapy. Post PET showed significant improvement in Anterior cingulate cortex (AC), left caudate head (CN), thalamus(T), medial temporal cortex (MT), cerebellum (C).
Discussion
Cerebral palsy refers to a group of neurological condition that affects the body movement and posture. CP patients primarily have motor impairment but many of them experience additional comorbidities such as intellectual disability, hearing loss, vision impairment, and epilepsy [13,14]. According to the Surveillance for Cerebral Palsy in Europe (SCPE) collaboration, 31% of children with cerebral palsy (CP) have severe intellectual disability, 11% have severe visual disability, and 21% have epilepsy [15].
Cell transplantation, due to the regenerative and reparative potential of the cells is being actively explored as a potential treatment alternative for neurological disorders such as CP, Autism, ID [8,16-24]. Different types of cells and routes of administration have been studied to address the multifactorial representations in children with CP and to gain the best possible clinical outcomes [25,26]. In this case, the patient was intrathecally administered with autologous BMMNCs. These cells are easily accessible, available in adequate quantity and associated with minimal risks [27]. The injected cells home onto the target tissue and restore the lost or damaged cell by inducing neural repair. They either carry out the repair process by directly differentiating into neural cells and replacing the lost ones or indirectly through paracrine mechanism.
The neurodeficits observed in CP are a result of the injury to the infant brain [28]. The cause of CP with ID in our case was hypoxic ischemic damage due to oxygen deprivation. One of the pathophysiology of CP is activation of microglia after hypoxic ischemic injury resulting in the production of proinflammatory cytokines and oxidative stress which triggers the excitotoxic cascade eventually leading to cell death [29]. Cerebral white matter injury is another common feature in CP resulting in loss of oligodendrocytes [30]. All these pathological mechanisms result in myelination defect, degeneration of the thalamus with cortical maldevelopment, gliosis and disruption of nerve conduction [31]. The defect in the connectivity of the neuronal network, especially in early childhood, limits the ability of the brain to process information leading to learning and intellectual disabilities and behavioral issues. Stem cells secrete neurotrophic factors which leads to neuroprotection, suppression of inflammation, angiogenesis and stimulation of endogenous neurogenesis. This improves the metabolism of the affected tissue and replaces the lost oligodendrocytes causing remyelination which in turn results in restoration of the neural connections thereby improving neuronal function [32,33]. Through angiogenesis, they improve blood and oxygen supply to the brain hence reversing the hypoxia.
The patient was on continuous rehabilitation regime post cell transplantation. Exercise has been shown to improve cognitive function in CP patients by mobilization of the stem cell and angiogenesis. It improves neovascularization and release of cytokines and nerve growth factors [34,35]. The combined effect of cellular therapy and neurorehabilitation resulted in improved motor functions, cognition, muscle tone, posture and gait supported by improved scores on objective scales.
PET imaging of the brain has shown to be a powerful monitoring tool for detecting neurofunctional abnormalities associated with various neurological diseases [36]. PET imaging, when combined with computed tomography (CT), provides information regarding metabolic changes at the cellular level in different regions of the brain [37]. The main objective to do PET CT scan in this case study was to measure the changes in metabolism which is associated with repair of the nerve tissue [11]. An increased metabolism indicates improved functioning [38]. A comparative PET CT scan performed before and after cell transplantation demonstrated improved metabolic activity in anterior cingulate cortex, left caudate head, thalamus, medial temporal cortex, cerebellum. The functional improvements demonstrated by the patient corresponded to the areas of the brain that showed evidence of improvements in metabolism (Table 1).
| Areas of the brain showing increased metabolism | Function improved |
|---|---|
| Temporal lobe | Memory, Social interaction, learning |
| Thalamus | Sensory integration |
| Cerebellum | Fine motor activities, voluntary control improvements and walking balance |
| Caudate head | Memory, learning |
| Anterior cingulate cortex | Decision making, emotion regulation |
Table 1: Areas of brain showing increased metabolism in the PET scan, carried out in six months after cell therapy corresponding to the functional improvement.
Limitations
This is a single case of CP with coexisting ID and independent effect of rehabilitation or cellular therapy cannot be distinguished. Significant functional improvements were seen after the cell transplantation combined with rehabilitation even at the age of 17 years. This suggests that cellular therapy has a crucial role in improving the neuronal function which was supported by the PET CT brain studies.
Conclusion
All clinical and objective changes in the patient suggest that the autologous bone marrow mononuclear cell transplantation along with neurorehabilitation is safe and effective therapeutic modality for the child suffering from CP and ID. It can give symptomatic, functional and cognitive improvements thereby improving the quality of life. Hence, it can be used as a supplement to the standard treatment for enhanced outcome.
Conflicts of Interest
The authors declare that there is no conflict of interest regarding the publication of this article.
References
- Mutch L, Alberman E, Hagberg B, Kodama K, Perat MV (1992) Cerebral palsy epidemiology: where are we now and where are we going? Develop Med Child Neurol 34: 547-551.
- Huang H, Chen L, Xi H, Wang Q, Zhang J, et al. (2009) Olfactory ensheathing cells transplantation for central nervous system diseases in 1,255 patients. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 23: 14-20.
- Miller G, Clark GD (1998) The cerebral palsies: Causes, consequences and management. Butterworth-Heinemann Medical, Elsevier, UK.
- Lagunju IA, Okereke J, Adebayo A, Eni-Olorunda T (2010) Neurocognitive and sensory impairments in cerebral palsy. J Pediatric Neurol 8: 385-390.
- Kolaski K, Logan LR (2008) Intrathecal baclofen in cerebral palsy: A decade of treatment outcomes. J Pediatric Rehabilitation Med 1: 3-32.
- Delgado MR, Hirtz D, Aisen M, Ashwal S, Fehlings DL, et al. (2010) Practice parameter: Pharmacologic treatment of spasticity in children and adolescents with cerebral palsy (an evidence-based review): Report of the Quality Standards Subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society. Neurology 74: 336-343.
- Gogel S, Gubernator M, Minger SL (2011) Progress and prospects: stem cells and neurological diseases. Gene Therapy 18: 1.
- Sharma A, Sane H, Gokulchandran N, Badhe P, Kulkarni P, et al. (2014) Stem cell therapy for cerebral palsy–a novel option. In Cerebral Palsy-Challenges for the Future. InTech.
- Sharma A, Sane H, Kulkarni P, Yadav J, Gokulchandran N, et al. (2015) Cell therapy attempted as a novel approach for chronic traumatic brain injury–a pilot study. Springer Plus 4: 26.
- Sharma A, Sane H, Gokulchandran N, Khopkar D, Paranjape A, et al. (2014) Autologous bone marrow mononuclear cells intrathecal transplantation in chronic stroke. Stroke Res Treatment 2014.
- Kannan S, Chugani HT (2010) Applications of positron emission tomography in the newborn nursery. In: Seminars Perinatology 34: 39-45.
- World Medical Association (2013) World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA 310: 2191.
- Novak I, Walker K, Hunt RW, Wallace EM, Fahey M, et al. (2016) Concise Review: Stem Cell Interventions for People with Cerebral Palsy: Systematic Review with Meta-Analysis. Stem Cells Translational Med 5: 1014-1025.
- Novak I, Hines M, Goldsmith S (2012) Clinical prognostic messages from a systematic review on cerebral palsy. Pediatrics 130: 285-312.
- Pruitt DW, Tsai T (2009) Common medical comorbidities associated with cerebral palsy. Physical Medicine and Rehabilitation Clinics of North America 20: 453-467.
- Sharma A, Sane H, Gokulchandran N, Kulkarni P, Gandhi S, et al. (2015) A clinical study of autologous bone marrow mononuclear cells for cerebral palsy patients: a new frontier. Stem Cells International 2015.
- Sharma A, Sane H, Kulkarni P, D’sa M, Gokulchandran N, et al. (2015) Improved quality of life in a case of cerebral palsy after bone marrow mononuclear cell transplantation. Cell Journal 17: 389.
- Sharma A, Kulkarni P, Sane H, Gokulchandran N, Badhe P, et al. (2012) Positron emission tomography-computed tomography scan captures the effects of cellular therapy in a case of cerebral palsy. J Clin Case Rep 2: 2.
- Sharma A, Geng T, Sane H, Kulkarni P (2017) Clinical neurorestorative progresses in cerebral palsy. J Neurorestoratol 5: 51-57.
- Sharma A, Sane H, Pai S, Kulkarni P, Raichur M, et al. (2017) Intrathecal Administration of Autologous Bone Marrow Mononuclear Cells in a Case of Cerebral Palsy Coexisting with Autistic Features. Phys Med Rehabil Int 4: 1110.
- Sharma A, Sane H, Paranjape A, Gokulchandran N, Kulkarni P, et al. (2013) Positron emission tomography-computer tomography scan used as a monitoring tool following cellular therapy in cerebral palsy and mental retardation-a case report. Case Rep Neurol Med 2013.
- Sharma A, Sane H, Pooja K, Akshya N, Nandini G, et al. (2015) Cellular Therapy: A Novel Treatment Option for Intellectual Disability: A Case Report. J Clin Case Rep 5: 483.
- Sharma A, Gokulchandran N, Sane H, Pai S, Kulkarni P, et al. (2017) Cognitive Changes after Cellular Therapy in a Case of Intellectual Disability. J Transplant Stem Cel Biol 4: 4.
- Sharma A, Gokulchandran N, Sane H, Nagrajan A, Paranjape A, et al. (2013) Autologous bone marrow mononuclear cell therapy for autism: an open label proof of concept study. Stem Cells International 2013.
- Fischer UM, Harting MT, Jimenez F, Monzon-Posadas WO, Xue H, et al. (2009) Pulmonary passage is a major obstacle for intravenous stem cell delivery: the pulmonary first-pass effect. Stem Cells Dev 18: 683-692.
- Kang SK, Lee DH, Bae YC, Kim HK, Baik SY, et al. (2003) Improvement of neurological deficits by intracerebral transplantation of human adipose tissue-derived stromal cells after cerebral ischemia in rats. Exp Neurol 183: 355-366.
- Lindvall O, Kokaia Z (2010) Stem cells in human neurodegenerative disorders—time for clinical translation? J Clin Investigation 120: 29-40.
- Gnecchi M, Zhang Z, Ni A, Dzau VJ (2008) Paracrine mechanisms in adult stem cell signaling and therapy. Circulation Res 103: 1204-1219.
- Tran C, Damaser MS (2015) Stem cells as drug delivery methods: application of stem cell secretome for regeneration. Adv Drug Delivery Rev 82: 1.
- Rosenbaum P, Paneth N, Leviton A, Goldstein M, Bax M, et al. (2007) A report: the definition and classification of cerebral palsy April 2006. Dev Med Child Neurol Suppl 109: 8-14.
- Marret S, Vanhulle C, Laquerriere A (2013) Pathophysiology of cerebral palsy. In: Handbook of Clinical Neurology 111: 169-176.
- Nakamura Y, Okudera T, Hashimoto T (1994) Vascular architecture in white matter of neonates: Its relationship to periventricular leukomalacia. J Neuropathol Exp Neurol 53: 582-589.
- Pocock JM, Kettenmann H (2007) Neurotransmitter receptors on microglia. Trends in Neurosciences 30: 527-535.
- Fabel K, Wolf S, Ehninger D, Babu H, Galicia P, et al. (2009) Additive effects of physical exercise and environmental enrichment on adult hippocampal neurogenesis in mice. Frontiers in Neuroscience 3: 2.
- Teng YD, Liao WL, Choi H, Konya D, Sabharwal S, et al. (2006) Physical activity mediated functional recovery after spinal cord injury: potential roles of neural stem cells. Regenerative Medicine 1: 763-776.
- Altman DI, Volpe JJ (1991) Positron emission tomography in newborn infants. Clin Perinatol 18: 549-562.
- Kato T, Nakayama N, Yasokawa Y, Okumura A, Shinoda J, et al. (2007) Statistical image analysis of cerebral glucose metabolism in patients with cognitive impairment following diffuse traumatic brain injury. J Neurotrauma 24: 919-926.
- Raichle ME (1994) Visualizing the mind. Scientific American 270: 58-64.

Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences